In my experience, timely intervention can make a difference. That’s why I consider initiating UPTRAVI® earlier when appropriate.
Mrinalini Krishnan, MD, FACC
Regional Director, Pulmonary Hypertension, Assistant Professor of Medicine, Advanced Heart Failure Cardiologist
MedStar Heart and Vascular Institute
Washington, DC
Dr Mrinalini Krishnan is a paid consultant of Johnson & Johnson.
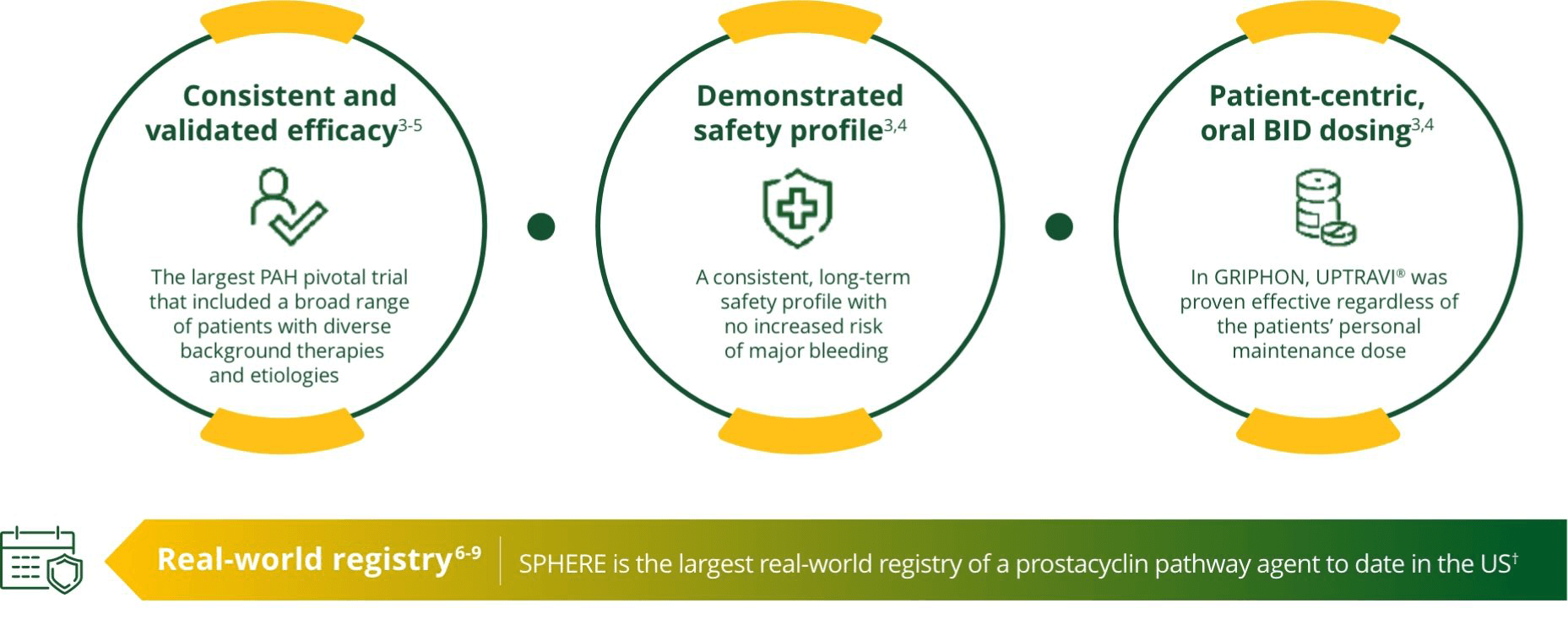
Target the Foundational Prostacyclin Pathway When Treating PAH1

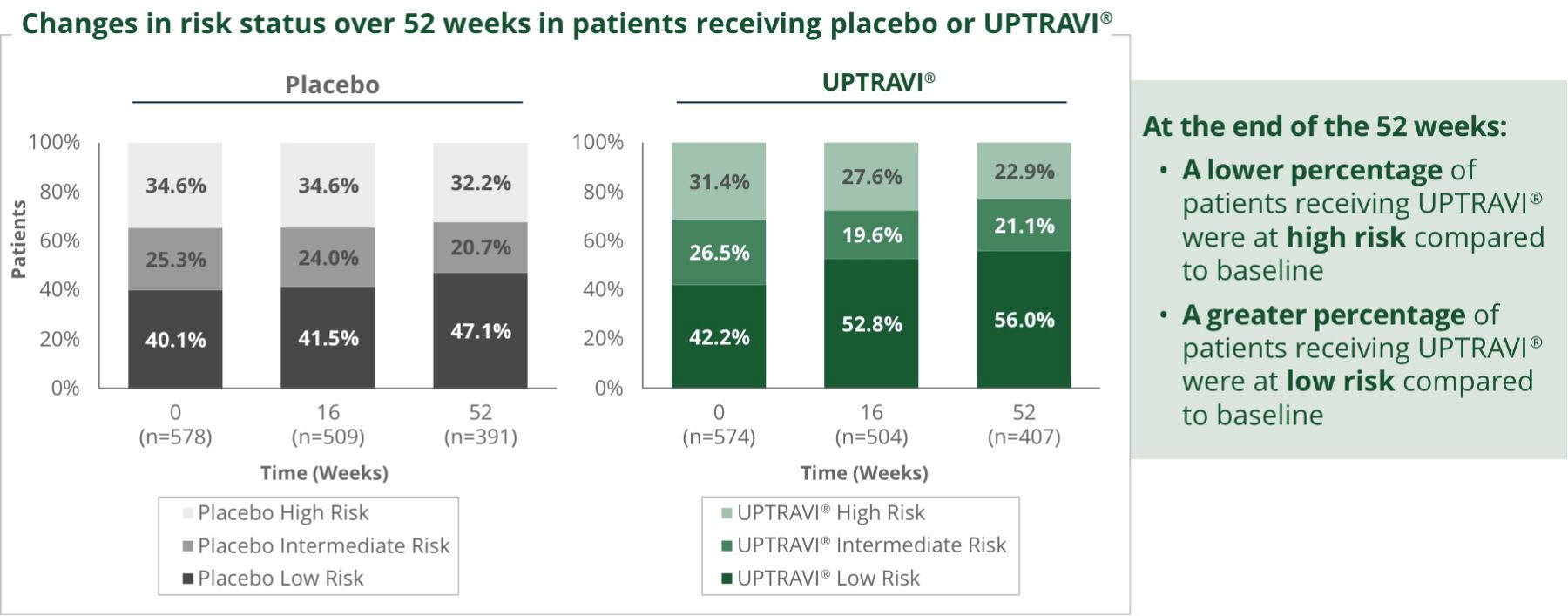
Based on a post hoc analysis of data from the prospective COMPERA registry (N=1655), an ongoing web-based PH registry launched in 2007 that collects baseline, follow-up, and outcome data from patients who receive targeted therapies for PH. Specialized centers in several European countries participate, with ~80% of the patients coming from German PH centers. In this analysis, patients were newly diagnosed with any form of PAH between January 1, 2009, and December 31, 2020. Risk assessment was evaluated at baseline and follow-up. At first follow-up, 17.3% (n=245) were classified as high risk, 37.8% (n=534) were classified as intermediate-high risk, 27.9% (n=395) were classified as intermediate-low risk, and 17% (n=240) were classified as low risk.2
The 2022 ESC/ERS Guidelines recommend adding UPTRAVI® for patients without cardiopulmonary comorbidities who are at intermediate-low risk despite receiving ERA/PDE5i therapy. In these patients, switching from a PDE5i to riociguat may also be considered. In patients who present as intermediate-high or high risk taking oral therapies, also consider adding UPTRAVI® or switching from a PDE5i to riociguat if it is not feasible to add IV or SC prostacyclin analogs.1
Class I recommendation definition: evidence and/or general agreement that a given treatment or procedure is beneficial, useful, effective. Level B evidence definition: data derived from a single randomized clinical trial or large nonrandomized studies.1
COMPERA=Comparative, Prospective Registry of Newly Initiated Therapies for Pulmonary Hypertension; ERA=endothelin receptor antagonist; ESC/ERS=European Society of Cardiology/European Respiratory Society; IV=intravenous; PDE5i=phosphodiesterase type 5 inhibitor; PH=pulmonary hypertension; SC=subcutaneous.
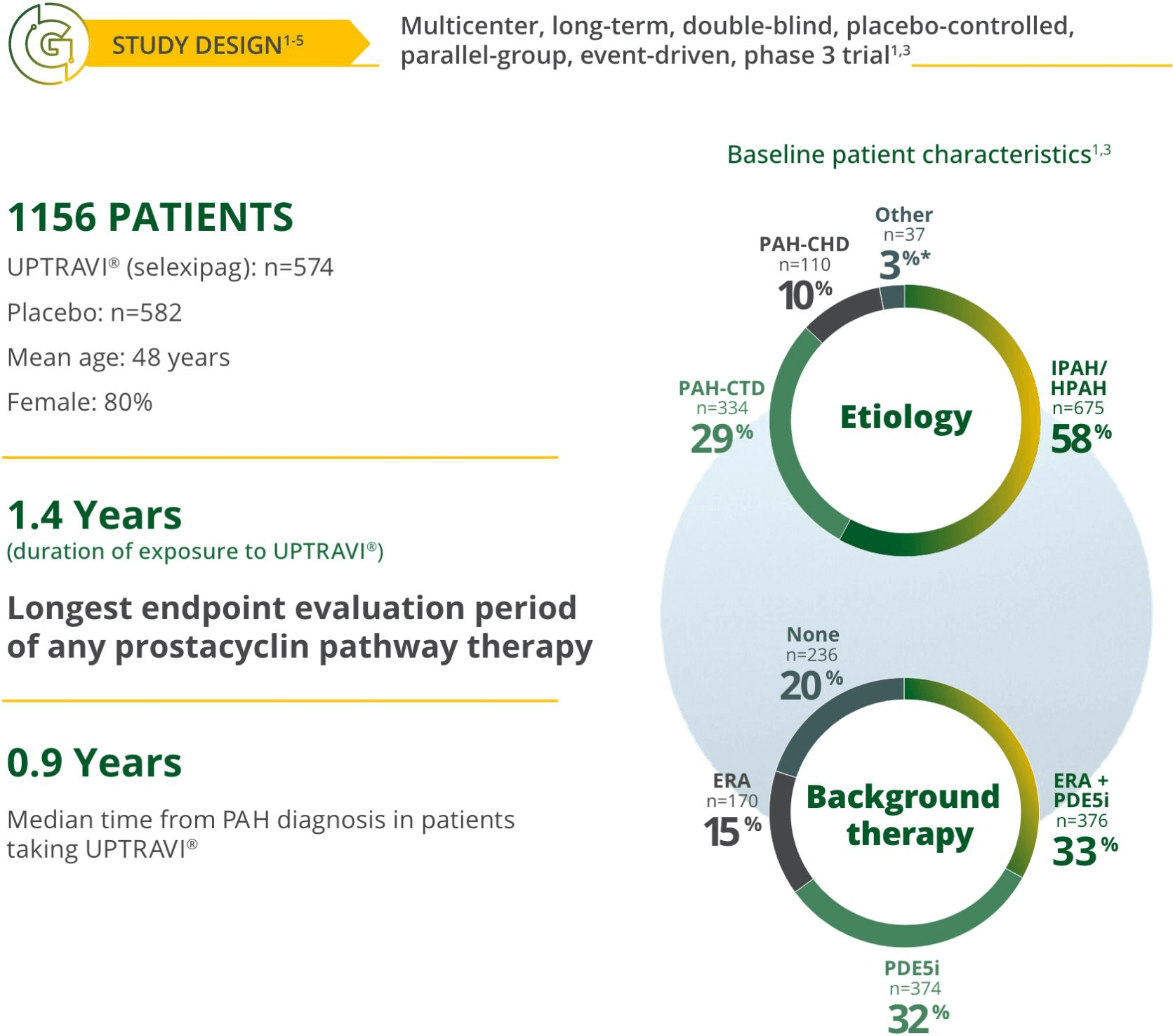
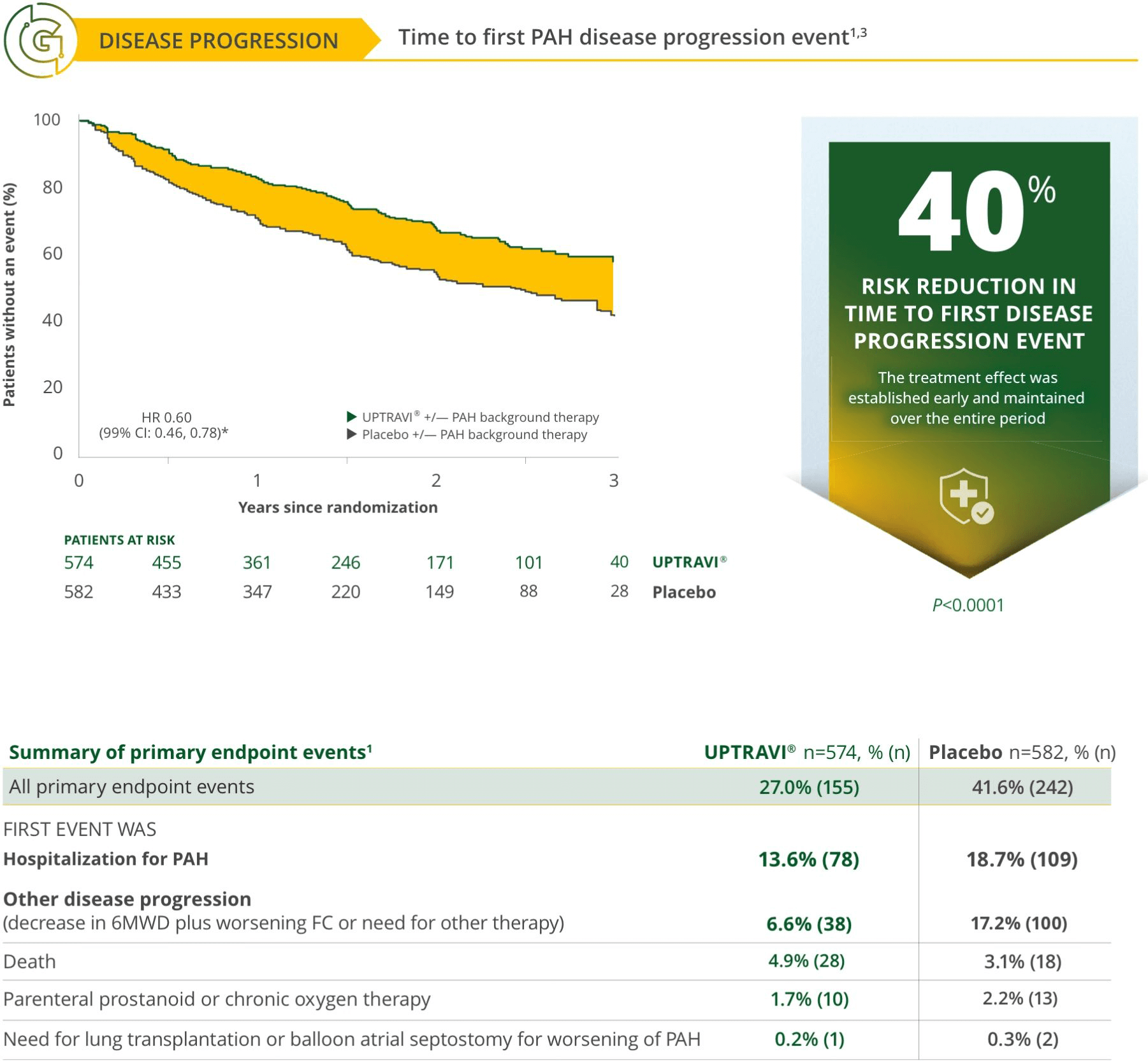
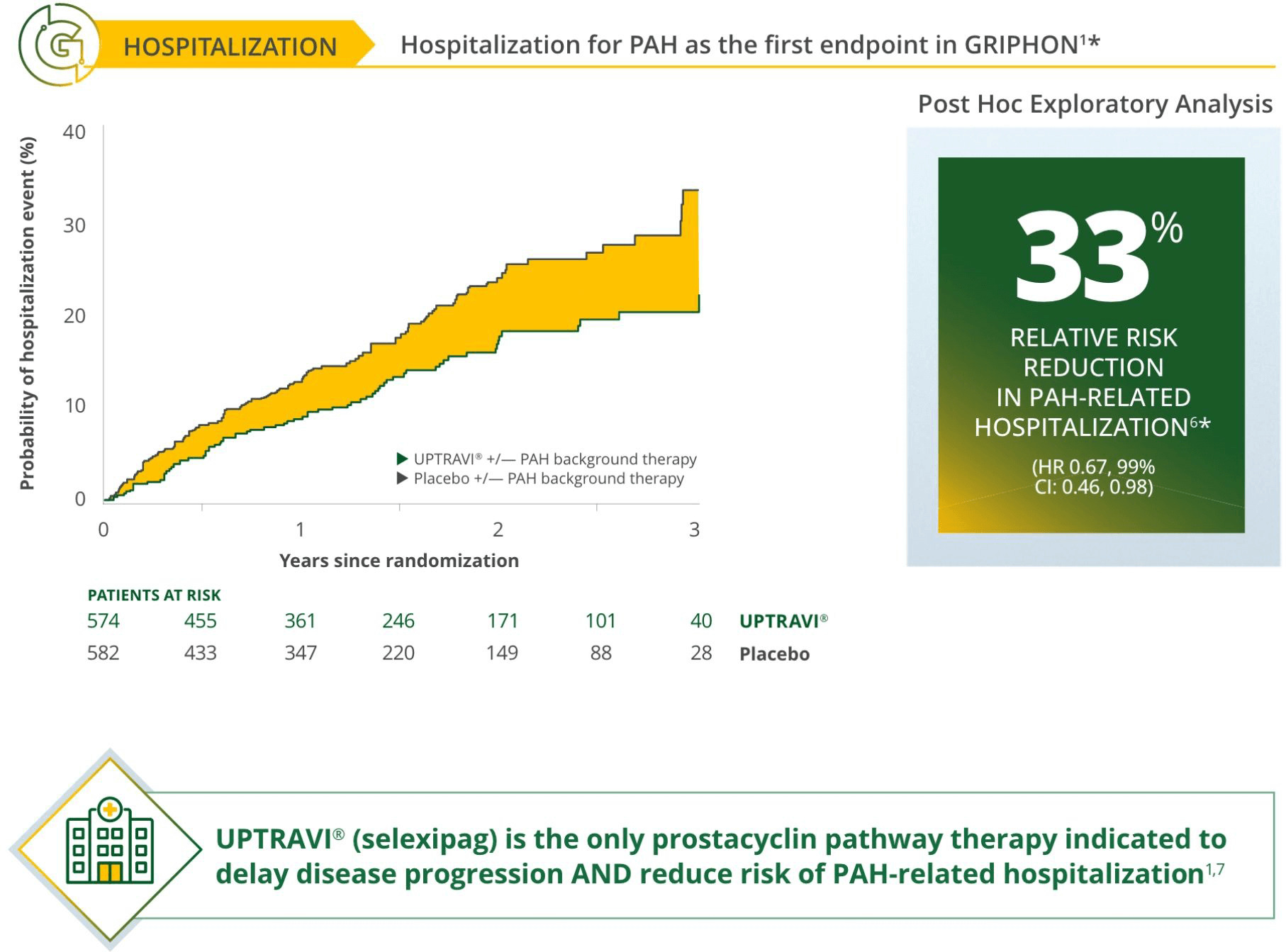
Treat early with UPTRAVI®3
2022 ESC/ERS Guidelines recommend UPTRAVI® at first follow-up for intermediate–low-risk patients1*
The 2022 ESC/ERS Guidelines recommend adding UPTRAVI® for patients without cardiopulmonary comorbidities who are at intermediate-low risk despite receiving ERA/PDE5i therapy. In these patients, switching from a PDE5i to riociguat may be considered. In patients who present as intermediate-high or high risk on oral therapies, also consider adding UPTRAVI® or switching from a PDE5i to riociguat if it is not feasible to add IV or SC prostacyclin analogs.
Based on published data as of January 7, 2025.
BID=twice daily (bis in die); IP=inositol phosphate; MOA=mechanism of action.
References: 1. Humbert M, Kovacs G, Hoeper MM, et al. 2022 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Heart J. 2022;43(38):3618-3731. 2. Hoeper MM, Pausch C, Olsson KM, et al. COMPERA 2.0: a refined four-stratum risk assessment model for pulmonary arterial hypertension. Eur Respir J. 2022;60(1):2102311. 3. UPTRAVI® (selexipag) full Prescribing Information. Actelion Pharmaceuticals US, Inc. 4. Sitbon O, Channick R, Chin KM, et al. Selexipag for the treatment of pulmonary arterial hypertension. N Engl J Med. 2015;373(26):2522-2533. 5. Data on file. Actelion Pharmaceuticals US, Inc. Length and Population Size of PAH Pivotal Trials Confirmation. June 2024. 6. McLaughlin V, Farber HW, Highland KB, et al. Disease characteristics, treatments, and outcomes of patients with pulmonary arterial hypertension treated with selexipag in real-world settings from the SPHERE registry (SelexiPag: tHe usErs dRug rEgistry). J Heart Lung Transplant. 2024;43(2):272-283. 7. Lachant D, Minkin R, Swisher J, et al. Safety and efficacy of transitioning from selexipag to oral treprostinil in pulmonary arterial hypertension: Findings from the ADAPT registry. Pulm Pharmacol Ther. 2023;82:102232. 8. Frantz RP, Schilz RJ, Chakinala MM, et al. Hospitalization and survival in patients using epoprostenol for injection in the PROSPECT observational study. Chest. 2015;147(2):484-494. 9. Evaluate Real-World Use of Next Generation Infusion Pumps to Administer Remodulin (EVOLVE). Clinicaltrials.gov identifier: NCT05060315. Updated January 16, 2025. Accessed January 22, 2025. https://clinicaltrials.gov/study/NCT05060315.